How useful is the data you are collecting? Data collection is the foundation of any data-driven approach; the right data must be collected accurately and in abundance. In some cases, like medical device testing, data collection and preservation are required for compliance with regulatory standards. Data collection and analysis of data are the basis of quality improvement. It is safe to say. Data collection is an essential element of any manufacturing business.
The Case for Automation
While there are multiple types of data that are important to understand and feed the quality improvement  engine, let‘s look specifically at process data and parametric data. For example, process data is recorded to indicate the pass, fail, or visited result of an inspection or test step. Parametric data is collected from measurement instruments, like a power supply measuring 5 Vdc, for example.
engine, let‘s look specifically at process data and parametric data. For example, process data is recorded to indicate the pass, fail, or visited result of an inspection or test step. Parametric data is collected from measurement instruments, like a power supply measuring 5 Vdc, for example.
Automated data collection is one good way to ensure that good data is gathered. Data gathering using an automated system ensures the following:
- No human operator bias or dependence
- No human resource labor cost
- No process interruption as data is collected electronically in parallel with the process.
- Fast and accurate
The dangers of not using an automated data collection system are numerous. Here are a few possible scenarios which result in sub-par product quality:
- Product moves through a manufacturing step without the process result getting entered into the Quality Management System database (QMS)
- The product which successfully passes a manufacturing step is entered into the QMS as a fail
- The product which fails a manufacturing step is entered into the QMS as a pass
- The product that did not go through a manufacturing step gets entered as a pass in QMS
Benchmark takes data collection very seriously. Overarching all of the manufacturing processes at Benchmark is an internally developed tool called the Process Feedback System (PFS). An entire paper could describe PFS and how it is used to manage the quality of products from before a build begins through the return of material goods.
Benchmark's TestLink System
To better integrate automated data collection into manufacturing processes, Benchmark developed a tool called TestLink to gather and distribute data between manufacturing processes and PFS seamlessly. This middleware works between various process step applications and the process data repository, ensuring that relevant, accurate data is available for production and process improvement activities.
From the name, one may believe the tool is relegated to test processes. Indeed that was the initial intent and implementation. Since its launch, however, TestLink has been applied to inspection steps, oven verification, pick and place machines, laser marking tools, certificate of compliance printers, ultrasonic welders, and more. Now the value of automating data collection is appreciated beyond test processes and across the Benchmark manufacturing activities. 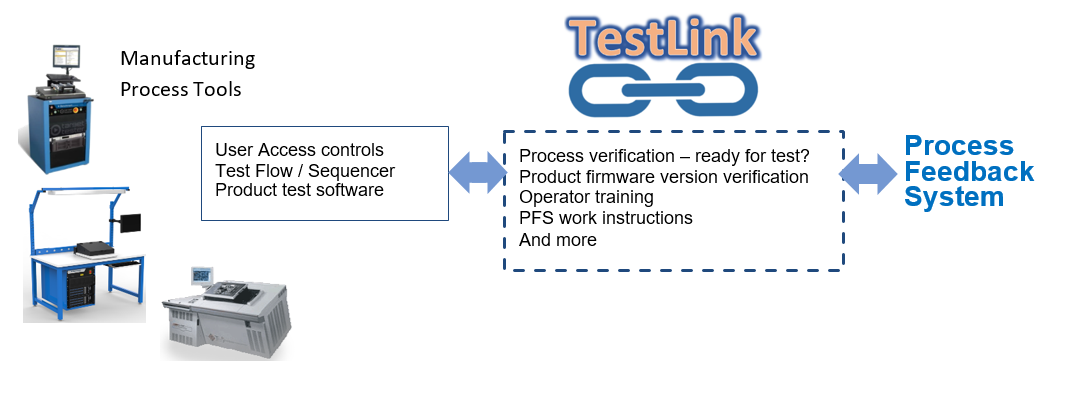
Leading in Process Improvement
Data is only as good as the process used to collect the information. Ensuring data is accurate and reliably collected is a goal of good manufacturing processes focused on product quality. Benchmark has a longstanding tradition of leading with creative solutions and designing the tools required for data-driven process improvement before those tools are available on the market. Indeed, the internally developed systems at Benchmark have received customer accolades and testimonials that our tools are better than those available commercially on the market today.
No matter how complex or for which purpose a test solution is required, Benchmark has the engineering expertise to start with a vision of the test need and to be able to drive the test design to completion. The advanced test design process we implement includes the requisite planning, design reviews, documentation, etc., which are expected of a mature design firm. Throughout it all, a foundation of reliable, relevant data drives performance and excellence in all we do.
Up next, I'll be tackling the topic of data storage for medical devices and then some of the woes of tester validation. Stay tuned for these and more topics relating to a test road map for a successful product launch.
Want to talk to Paul about your specific process data challenges? Contact us!
