As innovation in neuromodulation continues to expand the capabilities of neurostimulation technology, medical device manufacturers will be incentivized to optimize the lead interface designs they use in their new products. A change to what seems like a minor sub-assembly can make an immense difference in the product’s reliability and can save manufacturers significant time and money in bringing their product to market.
Manufacturing Roadblocks
Because of the proprietary nature of the leads used by different neurostimulator manufacturers, the lead interface often needs to be redesigned with every product, a costly and time-consuming aspect and manufacturing roadblock when bringing a new system to market.
The traditional approach of lead interface design in neurostimulation devices uses complex leaf-springs or fixed pad methods, which often require precision alignment techniques to manufacture and add significant process development time to the production schedule.
Other drawbacks to traditional lead interface designs include high lead insertion force, high electrode contact resistance and weakness during insertion and removal cycling, which create clinical challenges for both the surgeon and the patient.
A New Approach to Lead Interface Design
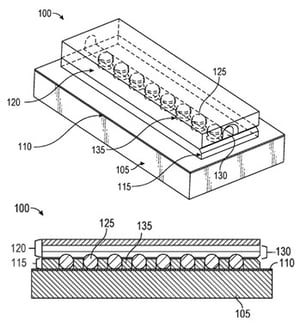 To overcome the challenges of the traditional approach, medical hardware manufacturers must consider new design methods. One such solution is an innovative, novel lead interface designed by the engineers at Benchmark, which utilizes spherical ball contacts that replace the complex leaf spring or fixed pad methods.
To overcome the challenges of the traditional approach, medical hardware manufacturers must consider new design methods. One such solution is an innovative, novel lead interface designed by the engineers at Benchmark, which utilizes spherical ball contacts that replace the complex leaf spring or fixed pad methods.
The new design can easily be configured for multiple lead types and shapes, while allowing for a more consistent manufacturing approach. And due to this highly configurable lead design, critical clinical parameters can be designed for, allowing ease of setup by the surgeon.
Leveraging Off the Shelf
As the technologies of neuromodulation continue to advance, I suspect that the ability to leverage ‘off the shelf’ solutions will quickly come to the forefront of manufacturers’ minds as we’ve seen with comparable technologies in the cardiac pacing market. While the applications of neurostimulators may seem limited today, it’s easy to imagine that in the near future each manufacturer will offer a suite of products that rely on the same core technology to treat a variety of ailments.
At the Leading Edge of Neuromodulation
Benchmark’s lead interface technology is patented, but available for use through licensing or other agreements. This innovative design allows customers to reduce process development time by utilizing a standardized set of components and optimizing standard assembly processes across multiple systems.
Reach out today to find out how you can leverage this new lead interface in your current or next generation device.
